
Resonance structures represent a Resonance Hybrid, which is a mixture of all resonance structures. the CNS molecule has the overall charge of 1- so, the FC of Lewis structure has to be equal to 1. 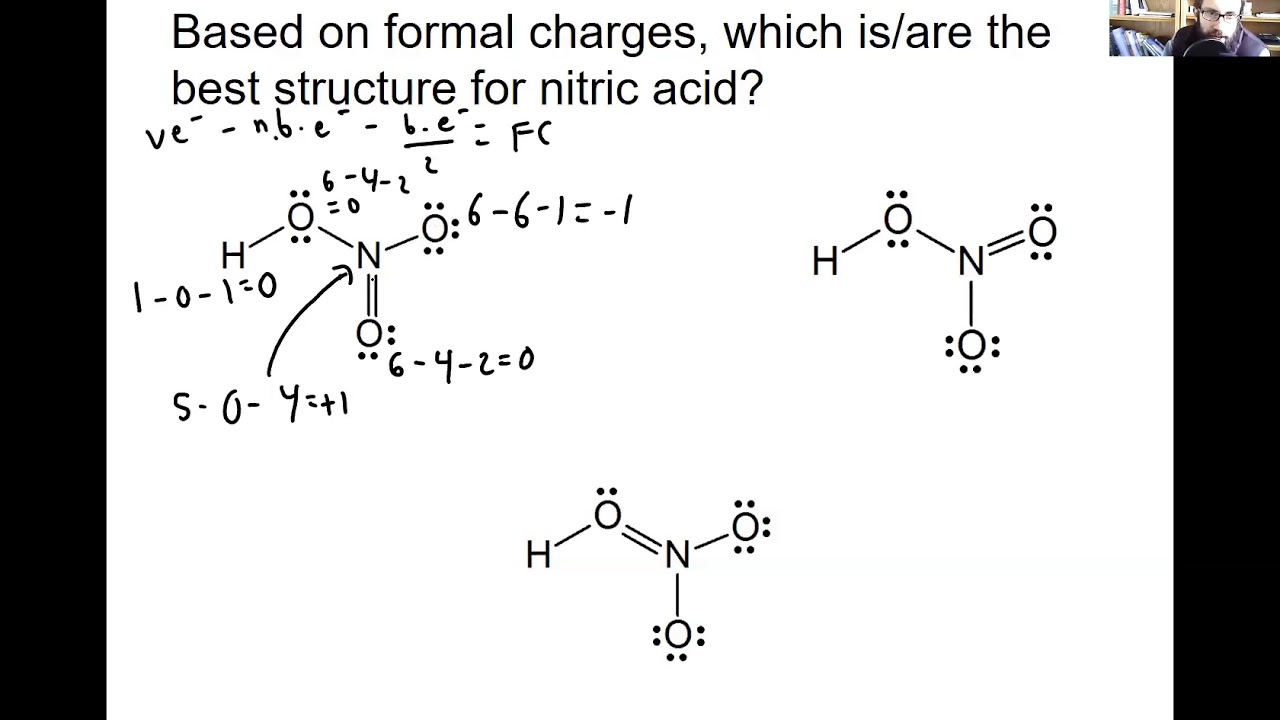
The FC has to be equal to the overall charge of the molecule, e.g. of valence electrons in free orbital) – (no. The formula to find Formal Charge is-įormal Charge = (no. We wish to select the resonance structure with the smallest formal charges, which adds to zero or the overall molecules’ charge. If calculated rightly, the molecule’s net formal charge should be identical to a molecule’s oxidation charge (the calculated charge while writing out the molecular and empirical formula). Formal charge refers to the charge that is allocated to a particular atom in a molecule. In resonance structures, the number of lone pairs should be the same.Īlthough the structures look alike, the (FC) formal charge might not be.The skeleton of a structure can’t differ to change (electrons can move only).The hybridization of a structure should remain the same.All resonance structures should be following the rules of drawing/writing the Lewis Structures.(Only check out the number of electrons by counting them). Resonance structures must consist of an identical number of (e−) electrons, no electrons should be added or subtracted.This movement of electrons is referred to as delocalization. Resonance Structures and Delocalization RulesĮlectrons have the capability to move for helping to stabilize a molecule in resonance structures. Lewis structure’s skeleton remains exactly the same, electron locations are only changed. Resonance structures don’t alter atoms’ relative positions. When there are two or more than two possibilities available, then the word resonance has its role to play. It’s just like keeping your hat either in your left hand or your right hand. The resonance forms describe the higher probabilities areas (electron densities). In atoms, molecules, and compounds, electrons don’t have fixed position but there’re probabilities to be found in some places (orbitals). A few of the resonance structures are more positive, favorable, or additionally supporting than others. This resonance hybrid represents the complete electron delocalization inside a molecule.Ī molecule containing many resonance structures is much more stable as compared to fewer resonance structures. The final sum of “valid resonance structures” is described as the resonance hybrid. Resonance structures or contributing structures are in use when a single Lewis structure considering only one molecule can’t completely illustrate the bonding which occurs in between the nearby atoms comparative to the “Empirical data” for the definite bond lengths present between the particular atoms. The Contributing structures are exactly not the isomers (an isomer is a molecule or ion with similar formula but different structure) of a target ion or molecule since they are only different considering the location of delocalized electrons. It definitely contrasts and compares two or more than two potential Lewis structures which are depicted by a specific molecule.Įach and every exclusive Lewis structure is known as a contributing structure (or arrangements, also diversely called resonance structure or canonical structure) of target ion or molecule. Here, the bonding is not particularly described out by a lone Lewis structure. Resonance dictates the method that is within the VB theory or Valence Bond Theory of bonding and the way of detailing the “delocalized electrons” within some molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
